
How to Source High Quality Pharmaceutical Chemicals Globally?
In the dynamic landscape of global trade, sourcing high-quality pharmaceutical chemicals remains a crucial challenge for many companies. Dr. Sarah Thompson, a leading expert in the pharmaceutical industry, emphasizes, "The integrity of drug formulation starts with the quality of the chemicals used." This quote encapsulates the essence of maintaining high standards in sourcing practices.
Pharmaceutical chemicals are the backbone of drug production. The importance of sourcing them from reliable suppliers cannot be overstated. Companies must navigate a complex web of regulations and quality assurance protocols. Identifying trustworthy suppliers can be daunting, especially in a global marketplace. There is a pressing need for thorough research and due diligence in this process.
Moreover, while many suppliers offer competitive pricing, quality must never be compromised. The risk of using subpar chemicals can lead to serious ramifications, including product recalls or health risks. Ultimately, balancing cost and quality is an ongoing struggle that demands vigilance and expertise in the pharmaceutical chemicals sector. In this pursuit, reflection on sourcing decisions is essential for maintaining high standards.

Identifying Reliable Pharmaceutical Chemical Suppliers Worldwide
Finding reliable pharmaceutical chemical suppliers globally is essential for maintaining quality. Start by assessing potential suppliers' certifications. Look for established certifications like GMP (Good Manufacturing Practice) that indicate compliance with high standards. Check their production facilities and quality control measures.
Networking can also reveal trustworthy suppliers. Attend international trade shows, conferences, or webinars focused on pharmaceutical industries. Engaging with professionals in the field builds credibility. Seek out feedback from peers who have previous experience with potential suppliers. Don't hesitate to ask for references and case studies.
Regular audits are vital. They enable you to gauge quality over time. Continuous assessment fosters long-term relationships. Suppliers may present well at a meeting but show inconsistent quality later. It’s crucial to adapt your sourcing strategy based on emerging data. Reflect on your experiences and remain open to new, reliable partners.
Evaluating Supplier Credentials and Certifications for Quality Assurance
When sourcing high-quality pharmaceutical chemicals, evaluating supplier credentials is crucial. Look for suppliers with certifications such as ISO 9001. This standard ensures that a supplier adheres to quality management principles. A certified supplier is likely to maintain a focus on quality control and continuous improvement.
Another important factor is the supplier's experience in the industry. Years of operation can indicate reliability and expertise. However, don’t assume that all long-standing suppliers are meeting modern standards. Ask for recent audits and compliance reports to ensure they keep up with regulations. Transparency in these documents builds trust and shows commitment to quality.
It's also wise to consider the geographical location of suppliers. Some regions have stringent regulations that contribute to quality. However, this does not guarantee that all suppliers from these areas are dependable. Always perform due diligence by checking their track record. Gather feedback from previous clients as it can highlight potential issues in their practices or product quality.
How to Source High Quality Pharmaceutical Chemicals Globally? - Evaluating Supplier Credentials and Certifications for Quality Assurance
| Supplier Location | Certification Type | Quality Assurance Standards | Production Capacity (kg/month) | Lead Time (days) |
|---|---|---|---|---|
| Germany | ISO 9001 | GMP, ICH | 5000 | 30 |
| United States | FDA Approved | cGMP, ISO 13485 | 3000 | 45 |
| India | WHO Certified | GMP, FSSAI | 7000 | 15 |
| China | ISO 14001 | GMP | 10000 | 25 |
Understanding Global Regulatory Standards for Pharmaceutical Chemicals

Navigating global regulatory standards is crucial for sourcing high-quality pharmaceutical chemicals. Different countries have unique regulations that significantly impact sourcing strategies. For instance, the World Health Organization (WHO) reported that approximately 10% of pharmaceutical products in low- and middle-income countries are substandard or falsified. This statistic underscores the importance of compliance with national regulations and quality assurance protocols.
Understanding the European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA) guidelines is essential. These organizations establish stringent requirements for drug manufacturing and quality control. They provide frameworks that ensure the safety and efficacy of pharmaceutical products. Companies must conduct thorough audits of potential suppliers to verify their compliance with these standards. Yet, relying solely on supplier claims can lead to oversights. It's vital to implement robust verification processes.
Documentation plays a pivotal role in compliance. Certificates of Analysis (CoA) and Good Manufacturing Practice (GMP) certifications are necessary. However, obtaining these documents from suppliers does not guarantee product quality. A proactive approach, including random sample testing and third-party verification, can help mitigate risks. Emphasizing traceability throughout the supply chain can further enhance reliability. The complexity of navigating these regulatory frameworks suggests that ongoing education and adaptation are vital for success in the pharmaceutical chemicals market.
Assessing Quality Control Processes in Chemical Manufacturing
When sourcing high-quality pharmaceutical chemicals globally, quality control processes are crucial. Observing these processes helps ensure that the chemicals meet safety and efficacy standards. Manufacturers should utilize stringent quality checks along the entire production line. This encompasses raw material selection to final product testing.
Effective quality control involves comprehensive testing methods. These methods may include analytical testing, stability studies, and impurity profiling. Each step must be well-documented and traceable. However, not all manufacturers may meet these rigorous standards consistently. Discrepancies can arise in testing methodologies and result interpretation. Hence, a thorough audit of a potential supplier’s quality control practices is essential before forming a partnership.
Furthermore, regulatory compliance is non-negotiable. Manufacturers must adhere to regional and international guidelines. These can be complex and vary significantly by location. In this environment, it is easy for manufacturers to cut corners. This could lead to serious consequences down the line. Establishing reliable communication with suppliers can mitigate risks. Asking detailed questions about their quality assurance processes is a vital step in sourcing effectively.
Establishing Effective Communication and Negotiation with Suppliers
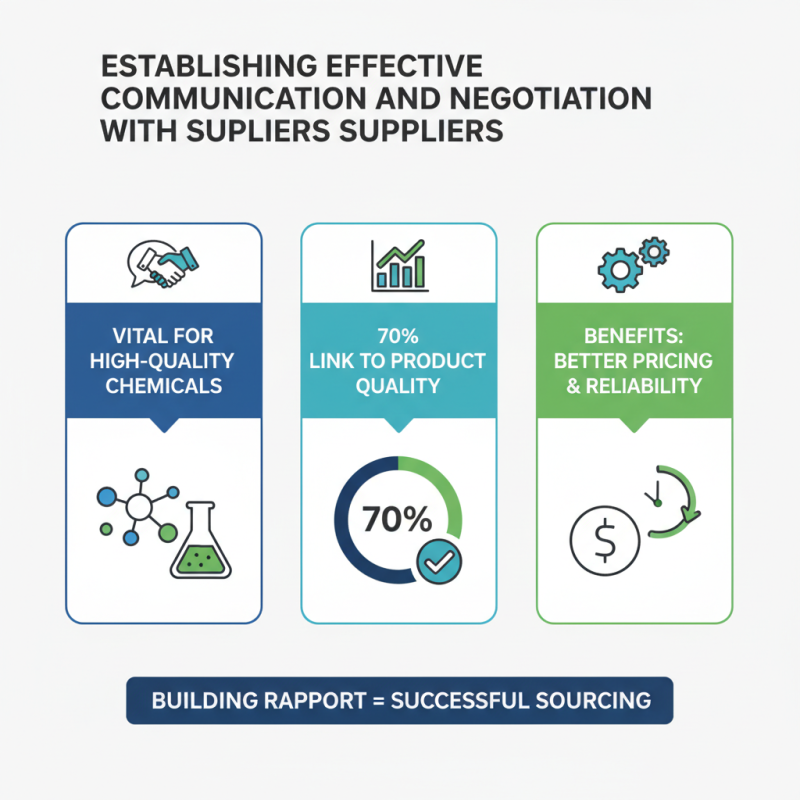
Establishing effective communication and negotiation with suppliers is vital in sourcing high-quality pharmaceutical chemicals. A recent survey revealed that about 70% of purchasing professionals consider supplier relationships crucial for product quality. Building rapport with suppliers can lead to better pricing and reliable delivery timelines.
Clear communication is central. This involves outlining specific requirements and expectations upfront. Using concise language helps avoid misunderstandings. Regular updates and feedback loops can also enhance collaboration. It's essential to cultivate trust, but this can be challenging. Some suppliers may overpromise their capabilities or downplay delivery times. Acknowledging these potential pitfalls is key.
Negotiation strategies should be data-driven. Research shows that companies that leverage analytics in procurement can achieve up to 15% savings on costs. Understanding market trends and supplier limitations empowers you during negotiations. For example, knowing regional supply constraints can help set realistic expectations. However, transparency is reciprocal; suppliers often appreciate honesty about your own limitations. This approach can lead to mutually beneficial outcomes, prompting long-term partnerships.
Related Posts
-
The Best Drug Raw Material Sources for Quality Pharmaceutical Production?
-
What Are the Uses and Benefits of Pharmaceutical Chemical Powder?
-

Essential Tips for Sourcing Pharmaceutical Intermediates?
-

Solutions for Sourcing the Best Pharma Materials in the Industry
-

7 Best Strategies for Sourcing High-Quality Pharmaceutical Intermediates in 2023
-

Unique Uses of Pharmaceutical Chemical Powder in Modern Medicine: Unveiling Real-World Applications
